Novo Nordisk's Ozempic® pill, the only FDA-approved oral peptide GLP-1 medication for adults with type 2 diabetes, soon to be available in the US
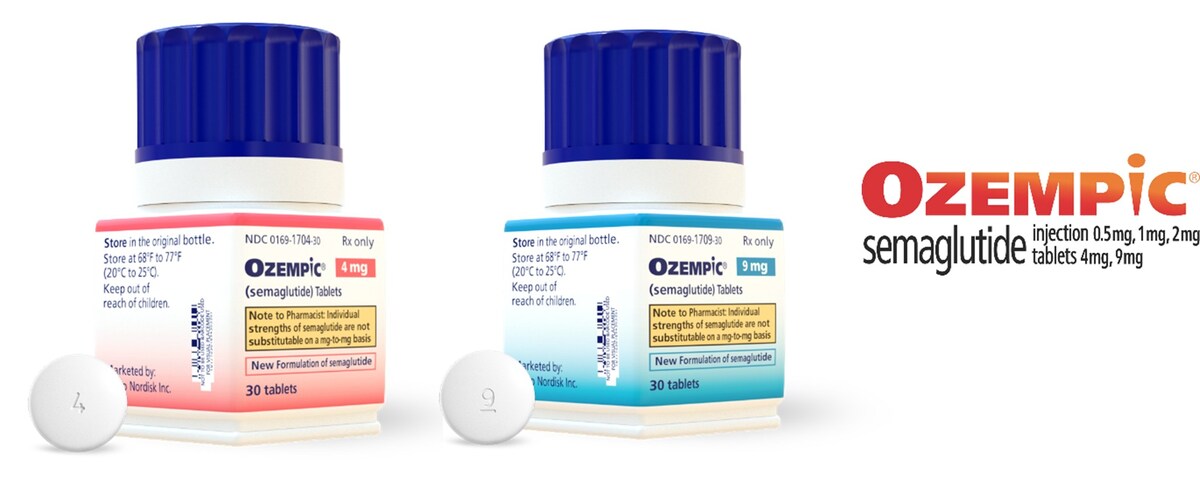 PLAINSBORO, N.J., May 1, 2026 /PRNewswire/ -- Novo Nordisk announced that starting Monday, May 4, Ozempic ® (semaglutide) tablets 1.5 mg, 4 mg, and 9 mg will be available for adults with type 2 diabetes in the US. Ozempic ® is the only FDA-approved oral peptide GLP-1 medication for adults with type 2 diabetes indicated not only to improve blood sugar, along with diet and exercise, but also to reduce the risk of major cardiovascular events (MACE) such as heart attack, stroke, or death in those who are also at high risk for these events. 1 Oral semaglutide for type 2 diabetes has been available as Rybelsus ® in a different formulation and set of doses.
PLAINSBORO, N.J., May 1, 2026 /PRNewswire/ -- Novo Nordisk announced that starting Monday, May 4, Ozempic ® (semaglutide) tablets 1.5 mg, 4 mg, and 9 mg will be available for adults with type 2 diabetes in the US. Ozempic ® is the only FDA-approved oral peptide GLP-1 medication for adults with type 2 diabetes indicated not only to improve blood sugar, along with diet and exercise, but also to reduce the risk of major cardiovascular events (MACE) such as heart attack, stroke, or death in those who are also at high risk for these events. 1 Oral semaglutide for type 2 diabetes has been available as Rybelsus ® in a different formulation and set of doses.
"Ozempic ® has become a leading, trusted brand for people with type 2 diabetes and the science is deep. Ozempic ® pill delivers strong blood sugar control and proven cardiovascular risk reduction, and people may also lose weight," said Ed Cinca, senior vice president of Marketing & Patient Solutions at Novo Nordisk Inc. "With Ozempic ® now available as both an injection and oral option under one brand, treatment can be individualized so patients are getting the proven semaglutide medicine that best fits their needs."
Ozempic ® pill is being manufactured end-to-end in the US and will be available through 70,000+ US pharmacies nationwide. To meet patients where they are, Ozempic ® pill will be available through self-pay options, such as NovoCare ® Pharmacy and select telehealth providers. This marks the first time semaglutide in a pill for type 2 diabetes is being offered through NovoCare ® Pharmacy.
The availability of Ozempic ® pill builds upon the well‑established heritage of Ozempic ® injectable medication, a familiar therapy widely used by people living with type 2 diabetes. Ozempic ® injection is a brand millions have been prescribed for the last eight years. Ozempic ® is the injectable peptide GLP-1 medicine with the most FDA-approved uses in type 2 diabetes, with indications to improve blood sugar along with diet and exercise, to reduce the risk of major cardiovascular events (heart attack, stroke, or death) in adults who also have known heart disease, and to reduce the risk of worsening kidney disease, kidney failure (end-stage kidney disease), and death due to cardiovascular disease in adults who also have chronic kidney disease. 1 Ozempic ® injection and Ozempic ® tablets are not approved for use in children.
Decades of diabetes research and the development of semaglutide have turned what many thought was impossible into a reality: delivering a peptide that could regulate specific biological functions within the body, in a pill for type 2 diabetes that mimics a naturally occurring hormone to lower blood sugar.
Semaglutide in pill form for type 2 diabetes was first approved by the FDA in 2019 as Rybelsus ® (semaglutide) tablets 3 mg, 7 mg, and 14 mg. The original strengths have been reformulated and are now being introduced as Ozempic ® (semaglutide) tablets 1.5 mg, 4 mg, and 9 mg. The new Ozempic ® pill works similarly to semaglutide tablets 3 mg, 7 mg, and 14 mg and has the same efficacy and safety profile as the original formulation offered in a smaller pill. 1-3 The product has been renamed to help patients and healthcare professionals more easily recognize the available treatment options for type 2 diabetes that contain the semaglutide molecule. Learn more about Ozempic ® pill at Ozempic-pill.com.
Ozempic ® pill is the only oral GLP-1 medicine approved to reduce the risk of heart attack, stroke, or death in adults with type 2 diabetes who are at high risk for these events across both primary prevention (lowering the risk of major cardiovascular events in people who are at higher risk) and secondary prevention (reducing the chance of another cardiovascular event in people who have already had one). 1,4
"Type 2 diabetes care isn't one‑size‑fits‑all, and how a medicine fits into someone's daily life really matters," said David Kayne, MD, CDCES, Partner at the Cedars-Sinai Medical Group, Encino, CA. "For some people, having a pill option can fit better into their daily routines. While injectable and oral medicines both treat adults with type 2 diabetes, they are not the same - and as clinicians, our goal is always to work with patients to find the option that best fits their needs, preferences, and routines."
Ozempic ® pill will be available to all eligible patients with multiple affordability options. Ozempic ® pill will be covered by insurance for most patients with type 2 diabetes for as little as $25 for up to a 3-month prescription. Self-pay patients using NovoCare ® Pharmacy or select telehealth providers will pay $149 for the starting dose of 1.5 mg Ozempic ® pills, $199 per month for the 4 mg Ozempic ® pills, and $299 per month for the 9 mg Ozempic ® pills. Government beneficiaries are excluded and eligibility and restrictions apply to this offer. One month is defined as one bottle of 30 tablets of Ozempic ® pill. Novo Nordisk reserves the right to modify or cancel this program at any time. See full terms at www.savingscardeligibility.com and NovoCarePharmacyTerms.com.
Rybelsus ® patients should continue their medication as directed and speak with their healthcare professionals about transitioning to the Ozempic ® pill, as appropriate. In advance of availability, Novo Nordisk is actively sharing clear, timely information with patients, caregivers, and healthcare professionals about the name change from Rybelsus ® to the Ozempic ® pill, its dosage strengths, and what people can expect at the pharmacy in May. Rybelsus ® will continue to be marketed outside the US.
Earlier this year, Novo Nordisk filed a supplemental drug application for Ozempic ® (semaglutide) tablets 25 mg for use in adults with type 2 diabetes. The company expects an FDA decision by the end of 2026.
Important Safety Information
Do not share your Ozempic ® pen with other people, even if the needle has been changed. You may give other people a serious infection or get a serious infection from them.
What is the most important information I should know about Ozempic ®?
Ozempic ® may cause serious side effects, including:
Do not use Ozempic ® if:
Before using Ozempic ®, tell your health care provider if you have any other medical conditions, including if you:
Tell your health care provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, herbal supplements, and other medicines to treat diabetes, including insulin or sulfonylureas.
What are the possible side effects of Ozempic ®?
Ozempic ® may cause serious side effects, including:
The most common side effects of Ozempic ® may include nausea, vomiting, diarrhea, stomach (abdominal) pain, and constipation. When taking Ozempic ® tablets, they may also include decreased appetite.
What is Ozempic ®?
Ozempic ® (semaglutide) injection 0.5 mg, 1 mg, or 2 mg and Ozempic ® (semaglutide) tablets 4 mg or 9 mg are prescription medicines used:
Ozempic ® injection is used:
Ozempic ® tablets are used:
It is not known if Ozempic ® injection and Ozempic ® tablets are safe and effective for use in children.
Please click here for Prescribing Information and Medication Guide for Ozempic ® (semaglutide) tablets. Please click here for Prescribing Information and Medication Guide for Ozempic ® (semaglutide) injection.
About Type 2 Diabetes
Type 2 diabetes is a chronic condition that affects how the body processes blood sugar (glucose) for energy. 5 According to 2023 CDC data, in the United States 40.1 million people have diabetes, with type 2 diabetes representing 90% to 95%, or an estimated 36 – 38 million people living with type 2 diabetes, making it the most common form of the disease. 5,6
About Novo Nordisk
Novo Nordisk is a leading global healthcare company that's been making innovative medicines to help people with diabetes lead longer, healthier lives for more than 100 years. This heritage has given us experience and capabilities that also enable us to drive change to help people defeat other serious chronic diseases such as obesity, rare blood, and endocrine disorders. We remain steadfast in our conviction that the formula for lasting success is to stay focused, think long-term, and do business in a financially, socially, and environmentally responsible way. With a US presence spanning 40 years, Novo Nordisk US is headquartered in New Jersey and employs approximately 10,000 people throughout the country across more than 10 manufacturing, R&D, and corporate locations in seven states plus Washington, DC. For more information, visit novonordisk-us.com, Facebook, Instagram, and X.
Contacts for further information
Media:
Liz Skrbkova (US)
+1 609 917 0632
[email protected]
Ambre James-Brown (Global)
+45 3079 9289
[email protected]
Investors:
Frederik Taylor Pitter (US)
+1 609 613 0568
[email protected]
References:
Novo Nordisk Inc.
US Communications
800 Scudders Mill Road
Plainsboro, NJ 08536
USA
Telephone: +1 609-987-5800
US26OZM00284
May 2026
SOURCE Novo Nordisk
